|
So how do you go about balancing an equation? These are the steps: First, count the atoms on each side. Altering the subscript would change the chemical composition of the molecule instead of the amount of molecules. 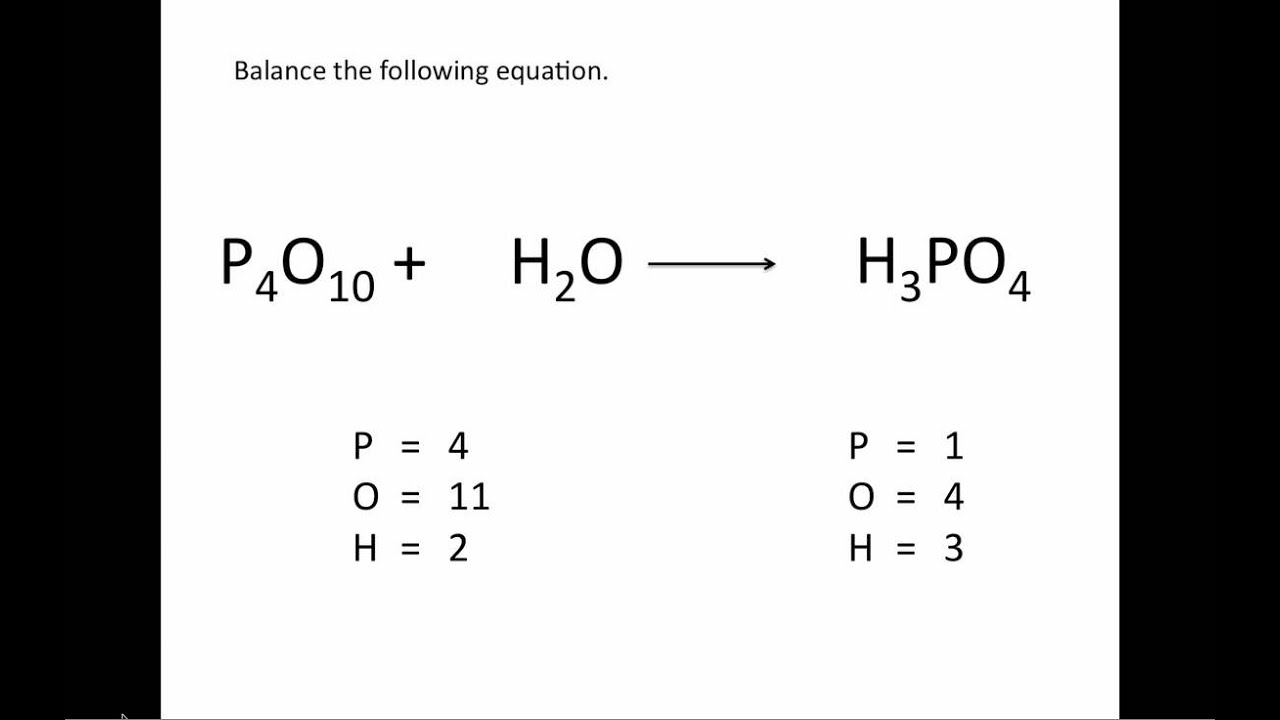
Balancing equations never involves changing the subscript of a molecule, only altering the coefficient. To determine the total amount of the atom present, multiply the coefficient by the subscript-the number of total molecules times the number of atoms in each molecule. For example, in 3O 2, the coefficient is 3 and the subscript is 2. The subscript represents the number of atoms of a given element in each molecule. The coefficient represents the number of molecules. Note that a coefficient, which appears to the left of a molecule, is different from a subscript, which appears in smaller print to the right of a molecule. Balancing an equation involves changing the coefficients-numbers placed in front of reactants or products to multiply them. 
This equation is already balanced because it has the same number of Ca and Cl atoms on each side. For example, consider the simple chemical reaction Ca + Cl 2 → CaCl 2. This means there must be the same mass of atoms on both sides of a chemical equation, and therefore the same number of atoms. The fundamental principle behind balancing equations is the law of conservation of mass, which states that matter, meaning physical substances like atoms and molecules, cannot be created or destroyed. Some examples of chemical reactions you might know include rusting, created by the reaction of iron with water and oxygen, and soda losing its bubbles, caused when carbonic acid splits into carbon dioxide and water. Reactants are the substances that enter a chemical reaction, while products are formed as the result of the reaction. The basis of chemical reactions is the idea that atoms and molecules frequently recombine into other molecules. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!īalancing chemical equations is a useful skill in chemistry.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
